This material is being copied from the original pamphlet and placed here for review and evaluation by members of Thunderbolts Forum. This is Copywrited material.
What Electricity Is
By James Roe
Chapter III
I HAVE GIVEN YOU a sketch of my thoughts and actions leading up to my discovery of "What Electricity Is," and a brief outline of the history of this great power. Now I will state my claims:
It is my finding that Electricity is Oxygen in an Energized Form.
There are those of you who will catch your breath and say "Bosh," but let me warn you, don't be too hasty with your laughs. First check the facts and reasoning that I will give you and then apply the information disclosed to any problem you have in electricity and see if you can answer, or solve them using these findings as a basis. If you can't then you can complain, if you so desire.
It is not my thought that my findings completely cover this subject, for it is vast in extent, but it is my belief that my findings give basic facts that will serve as a basis for many new discoveries in this and kindred fields.
You will find a number of repetitions in my work, but this is intentional for it adds emphasis to my points.
Following you will find facts and reasonings proving and supporting my findings.
PROOFS OFFERED
IN SUPPORT OF MY FINDINGS
Chapter IV
The Lodestone
I PRESUME that the first electricity ever handled by man was the charge contained in a lodestone, or magnetite.
It is a crystalline compound of oxide of iron from which the best Swedish steel is made. It is a natural, true magnet, having a positive and a negative pole located at opposite ends of the stone. There is an electronic field surrounding it which is typical of all true magnets.
Kingzette says the formula is (FeO, Fe2O3) or Fe3O4.
Now let me see if I can prove to you that Oxygen, in its energized form, is electricity.
It has been proven by many men that all types of electricity are the same, except that it varies in activity, presure, wavelength, volume and direction. This being true, if it is proven that Electricity is Oxygen in the lodestone then it proves that all Electricity is Oxygen. But don't worry, I won't only prove it in this way, with lodestone, it will be proven to you in several ways.
As I have said, the lodestone's formula is (FeO, Fe2O3). It is magnetic, it has a north and south pole and an electronic field surrounding it. Now let us reduce this substance, taking all of the oxygen from it, leaving pure iron (Fe). Now let us test the lodestone properties. We find that it has none. The poles are gone, the electronic field is gone. Its power to attract and repel is gone. The electric charge is gone.
From this experiment we find that when the Oxygen is removed from the lodestone (magnetite) the electric charge, or electron charge,
is removed, or:
We have removed an electric charge from an oxide of iron by taking away the Oxygen now let us see what happens when we add Oxygen to pure iron.
CHAPTER V
Oxy-Acetylene Cutting of steel
NOW LET US SEE what happens when the previously described action is reversed.
In the metal trades they make a great deal of use of a process that is absolutely the opposite of the experiment that was made with the lodestone. Instead of Oxygen being taken from a magnet, or magnetite, leaving pure iron without an electrical charge, in this experiment they add pure Oxygen to pure iron (Fe). And what do you suppose they get?
By adding pure Oxygen to pure iron at about 900 degrees C. the substance magnetite is formed. (FeO, Fe2O3).
The process wherin this action takes place is called Oxy-acetylene cutting, or burning of steel. The temperature of the iron is raised by contact with an oxy-acetylene flame until it reaches about 900 degrees C. and then a stream of pure Oxygen is poured into the hot metal. (Steel is hardened iron.) The Oxygen combines with the iron and the magnetite is formed.
Now let us test this magnetite (lodestone) for magnetic properties. It has a north and south pole, it has an electronic field, it attracts and repels, it has an electric charge. It is a true magnet.
This, as the previous experiment, has been proven many time so they are facts. These experiments prove that when you add or take away Oxygen you add or take away the Electric Charge. This proves the action to be reversible.
To many people these two experiments will be sufficient proof of my claim, but I must prove my findings to the most skeptical, they must be convinced that Electricity = (E/O), ENERGIZED OXYGEN, so let us continue and look into the different types of batteries used by us to produce electricity and see if I can produce more proof for you.
Primary Batteries
THE ACTION of the elements in my findings is contrary to the theories which are accepted by many, but this does not change my proof that ENERGIZED OXYGEN IS ELECTRICITY. Experiments performed by many of you will soon prove which is right and what the true action is that takes place.
In all primary batteries which require the replacement of parts after their charge is used up consists of two poles of different electrical conductivity, usually zinc (electrical conductivity 28) and carbon (electrical conductivity 0.2) placed in contact with some other compound containing Oxygen. If a conductor wire, usually copper, is connected from one pole to the other an electric current immediately starts to flow through the conductor from the Negative Pole to the Positive Pole.
As soon as the oxygen in the compound (it may be either liquid or solid) is used up the battery is said to be dead. No more electricity comes from the poles. The only way to revive it is to replace the parts. These batteries are so made that it is usually more economical to throw the battery away and get a new one.
Here again we have found that it is the Oxygen that forms, or as I claim is electricity.
CHAPTER VII
Secondary or storage Batteries
LET US TAKE a few minutes to look into the action which takes place in the secondary batteries, commonly known as storage batteries.
These batteries are usually a group of cells connected together to get the desired voltage, but each cell is identical. Each of these cells contains two poles, a positive and a negative pole. Both poles are made of lead and connect directly with lead plates constructed in the form of a grill. the positive grill plates are filled with lead peroxide (PbO2), a dark brown substance. The negative plates are filled with spongy lead which is greyish in color. These plates are immersed in an electrolyte which is usually dilute sulfuric acid. This combination of ftwo poles with an unequal power of conduction immersed in an electrolyte constitutes the simlpest kind of a voltaic cell.
As the battery comes into use chemical action takes place, and a discharge of electrons takes place, or you might say results
What happens?
It is written by others that these chemical changes cause the two plates, both positive and negative, to become converted to lead sulphite, and acid becomes more dilute. (This means that less (SO4) remains in the electrolyte.)
Let us look further into this action.
The positive plates are lead grills filled with lead peroxide. (PbO2)
The negative plates are lead grills filled with spongy lead.
Although both plates are made of lead (not metals of different conductivity) it must be realized that the spongy lead with which the negative grill is filled makes it capable of attracting many more electrons (electrons are the smallest division of electricity) than the positive plates which are filled with lead peroxide (PbO2). This is what makes the difference in the plates or/and poles in this type of battery.
As the two poles (plates), the positive plates filled with PbO2 and the negative plates (filled with sponge lead, making it the best conductor) are connected by a conductor, say a copper wire, chemical action starts up immediatly in the storage battery.
The negative plates, being the best conductor of electricity, attracts the electrons to it and the SO4 from the sulfuric acid electrolyte is deposited on the plates. The taking of the SO4 onto the plates and the hydrogen going off as gas means that some of the sulfuric acid has been removed from the electrolyte and it therefore is naturally more dilute.
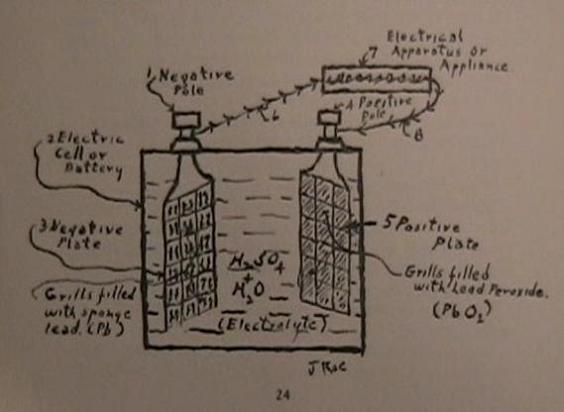
(Note--My illustrations are not intended to be proportionally, nor scientifically correct. Their only purpose being to simplify your understanding of my findings.)
My illustration shows a cross sectional view of a storage battery, but showing only one negative plate and one positive plate, with their respective poles. The plates are turned at and angle to show the grill construction.
In the illustration we find at the left at 1, a negative pole, extending out of the top of an electric cell, 2. This pole is connected to a negative plate,3, which is submerged in the electrolyte, dilute sulfuric acid. On the right, at 4, we have the positive pole connected to the positive plate, 5, which is similarly located, but seperated from the negative plate. At 6 we have a conductor wire, leading from the negative pole to an electrical apperatus, or appliance, 7. From this appliance we have a conductor, 8, returning to the cell and connected to the positive pole, 4.
As soon as the conductor wires are connected a current begins to flow. (It should be noted here that electrons show no activity unless they have a place to go. They must have a circuit to flollow.) The O2 from the PbO2 (lead peroxide) on the positive plate is replaced by the SO4 from the electrolyte. This O2 from the positive plate becomes 2 (E/O)(electrons) and passes over the negative plate and entering it as electricity it passes through the negative pole and the negative conductor to the apparatus. The Hydrogen from the sulfuric-acid (H2SO4) is given off as gas. SO4 also collects on the negative plate. As each molecule of SO4 is removed from the electrolyte it becomes more dilute, as should be expected. The nearer the battery comes to being discharged the more dilute the electrolyte becomes.
As the (e/o) (electrons in motion) issue from the negative pole it is electricity. It follows along the conductor until it reaches the apparatus. When it enters the apparatus it is under a certain pressure, and of certain volume. In operating the apparatus a certain portion of the electrons (electricity) are used (not destroyed), those not used are forced back into the return conductor and are returned to the positive pole of the cell. These electrons naturally have a lower pressure and a lesser volume and are known as positive electricity. If, when this positive electricity is returned to the cell, it cannot combine chemically with the contents thereof, it is given off as oxygen gas from the positive pole (plate).
As the available (E/O) in the battery lessens, the strength of the cell wanes, and so it should be, for (E/O) is ELECTRICITY, or ELECTRONS.
Again we have found that it is Oxygen that amkes, or as I claim is Electricity, but now let us see what happens when we recharge this battery.
To recharge a storage battery, it is connected up with a source of direct electricity, or electrical current. It is connected so that the electrons flow into the cells in a reverse direction from that taken by the electrons when the battery is discharging. The negative conductor from the charger is connected to negative pole of the battery and the positive to the positive. As the electrical current,(E/O), flows into the battery, chemical changes immediately start to take place, but these changes are exactly opposite to those which took place when the battery was being discharged. The sulphate (SO4) is, you might say, chased back into the electrolyte from whence it came, raising the concentration back to its original strength. The lead peroxide (PbO2) forms on the positive plates again and the negative plates are bare except for the sponge lead filling.
The storage battery has proven to us that when oxygen is taken from its contents, by chemical, electrical current results, and that when electric current, (E/O) (electrons) is returned to the battery it manifests itself or shows itself as Oxygen.
When electricity is producted by chemical action such as takes place in a storage battery, the reduction of the Oxygen in the chemicals involved is in direct proportion to the amount of current produced. The amount of current a battery is capable of producing is governed by the amount of (E/O), ENERGIZED OXYGEN, that can be freed from the chemicals contained in the cell.
The amount of (E/O) in a cell, within certain limits, can be varied at will by discharge, or recharge, of electrons.
In my findings on the storage battery it is apparent that the electrons, (E/O), rush to the plate filled with sponge lead, and thus the current, the electrons, in motion, electricity leaves the battery by this pole. The charge of (E/O) is negative and it leaves the battery through the negative pole. This is contrary to the theory of others, but read the balance of these writings before you make any definite opinions, yes read it several times and study it thoroughly for you will find much food for thought, and many argumentative pointsm but if you study it closely enough you will find the answers to most of your questions that may arise from your first reading, and also answers to many of the questions that may come up in your vocation, or avocation.
Regardless of whether any of my theories are proven to be right or wrong, there are sufficient FACTS disclosed in my findings to prove my claims, ELECTRICITY = ENERGIZED OXYGEN.
There are some that write that the terms denoting the charges of electricityy signify that one body is charged to a higher pressure that the other and that the one with the higher charge is termed positive and the one with the lower charge is termed negative. Again this is just opposite to my findings. Again I ask that you wait until you have studied by findings, and become thoroughly familiar with them before you voice an opinion. Use the information I have given you in these works, then add on your own knowledge, and your power of reasoning and see if you reach the same conclusions as I have.
If you can add anything to my proofs, or if you can disprove my findings that (E/O), ENERGIZED OXYGEN, OXYGEN IN AN ENERGIZED FORM IS ELECTRICITY, I would like very much for you to write me, giving me your opinion and arguments.
We have read something in the previous paragraphs about electrolytes but let us look at the next chapter and see what other interesting facts we can uncover in our quest for supporting evidence to myclaim that ELECTRICITY IS (E/O).
Electrolytes
AN ELECTROLYTE is a substance capable of carrying an electric current, that will decompose into portions, cations (positive ion) and anions (negative ions).
The electrolyte used in the primary cells is usually solid substances, in most storage batteries the electrolyte is fluid.
In all electrolytes used in batteries which produce or store electricity (electrons), I wish to draw your attention to the fact that they either contain Oxygen, or are capable of obtaining and/or freeing it from some source, for without Oxygen no Electricity can possibly be produced, for (E/O), ENERGIZED OXYGEN IS ELECTRICITY.
The Oxygen in these electrolytes is stored up energy, (E/O), or Electrons, which is known as the current, the electric energy, the EMF (electromotive force) according to my findings
Both salts and acids my be used as electrolytes, but their power to produce Electricity, I repeat, depends upon their ability to FREE (E/O)(ENERGIZED OXYGEN) from available compounds whereupon the FREED OXYGEN, (E/O), becomes an ELECTRIC CHARGE.
Again I ask you can my claim be wrong?
Let us go now to Ozone and see what light this active substance can add to our solution.